What is it?
Tuberculosis (TB) is caused by an infection with the bacteria Mycobacterium tuberculosis. In rarer cases, TB may be attributed to infection with Mycobacterium bovis or Mycobacterium africanum.
TB is categorised as being an active disease (the infected person is displaying symptoms and is infectious) or a latent infection (the bacteria is dormant and the infected person has no symptoms and is not infectious). Latent TB can become active if an infected person’s immune system becomes weakened (due to a medical condition or taking immunosuppressive therapies), following serious illness, with drug or alcohol abuse, or with increasing age.
What to look for
Approximately 60% of TB cases in Australia present as pulmonary (lung) TB. Common symptoms include a chronic cough which may be accompanied by haemoptysis (coughing blood) as well as fevers, night sweats, weight loss and malaise (generally feeling unwell).
In some cases, infection can also occur in the brain, bones, kidneys or lymph nodes. TB meningitis (brain infection) and TB septicaemia (blood infection) are considered the most serious manifestations of TB disease.
Most people infected with TB are asymptomatic. 10% of people infected with TB will go on to develop clinical disease at some point in their lifetime.
How is it transmitted?
Pulmonary TB can be spread to others via aerosol transmission of infected droplets through coughing or sneezing. People with TB infections without lung or laryngeal involvement generally cannot transmit disease. However, urine is considered infectious in the case of renal TB. In countries where Mycobacterium bovis is prevalent (it is not prevalent in Australia), the bacteria can also be transmitted by ingesting unpasteurised milk.
A person is considered infectious until 14 days after being on active treatment AND until they have tested negative on a sputum sample. Following an active infection the bacteria will remain dormant (non-infectious) within scar tissue where it can be reactivated later in life (even decades later). Once reactivated the bacteria is capable of being transmitted again.
Epidemiology
Australia has one of the lowest rates of TB in the world, affecting less than 7 per 100,000 people. Aboriginal and Torres Strait Islander people residing in the Northern Territory and Far North Queensland have a much greater burden of disease compared with non-Indigenous people in those areas and Aboriginal and Torres Strait Islander people living in other states.
Globally, it is estimated that approximately one-quarter of the world’s population is infected with TB, causing 1.5 million deaths annually. Bangladesh, China, India, Indonesia, Nigeria, Pakistan, Philippines and South Africa have the highest incidence of infection, accounting for almost half of the world’s TB cases.
Children under 5 years of age, the older population, those with immunocompromise (due to medical condition or immunocompromising therapies) and those impacted by poor living conditions have the highest rates of infection.
Prevention
Vaccination with the BCG vaccine is effective in reducing the incidence of TB meningitis and death in children less than 5 years of age in countries where TB is prevalent. Vaccination is not routinely recommended in developed countries like Australia where disease incidence is low. In Australia the following groups of people are recommended to be vaccinated:
- children < 5 years living in Aboriginal and Torres Strait Islander communities in Queensland
- Aboriginal and Torres Strait Islander neonates living in Queensland
- children < 5 years of age travelling internationally to areas where tuberculosis is prevalent.
BCG vaccination for individuals travelling to areas with a high incidence of disease is ideally administered 4-6 weeks prior to travel.
Tuberculin skin testing
Tuberculin skin testing (TST) or Mantoux testing prior to BCG vaccination is recommended on a case-by case basis to determine if a person already has a level of immunity to TB. It involves the intradermal injection of a tuberculin purified protein derivative (PPD).
In people who have previously received a BCG or have previously had TB exposure, a hypersensitivity reaction can be recognised 48-72 hours later.
It is important to note that TST results may be unreliable for 4-6 weeks following a measles infection or receiving a measles-containing vaccine.
Vaccine administration
BCG vaccine is administered via the intradermal route. Only healthcare providers trained in intradermal technique should provide BCG vaccination.
The recommended site for injection is on the left arm over where the deltoid muscle inserts into the humerus. Administration at this site will minimise the risk of keloid scarring.
- In children < 12 months of age the recommended dose is 0.05ml
- In children > 12 months of age the recommended dose is 0.1ml
Other injected live-attenuated vaccines can either be administered on the same day as BCG or 4 weeks apart. There is no recommended interval between BCG and oral live-attenuated vaccines.
Contraindications and precautions
BCG is a live-attenuated vaccine and is therefore contraindicated in individuals with immunosuppression or in those who are pregnant.
If active eczema, dermatitis or psoriasis is present at the site of injection, vaccination should be deferred until the skin can be treated and is clear of symptoms.
Side effects following vaccination
BCG, like all vaccines, has a list of common and expected side effects and a list of rare side effects that may occur in the weeks following vaccine administration.
Common and expected side effects can include:
- a small red papule will appear at the injection site in the weeks following the vaccine
- an ulcer (open sore) may develop 2-3 weeks later (usually less than 1 cm in diameter) and last from a few weeks to months
- the majority of infants will develop a flat scar at the site once the ulcer heals.
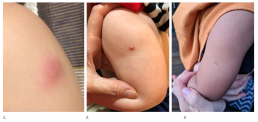
1. Papule
2. Ulcer
3. Scar
Rare or more serious side effects can include:
- axillary lymphadenopathy (swelling of the lymph nodes under the left arm)
- persisting ulcer lasting longer than a few months
- a large abscess (collection of pus) at the injection site
- keloid scarring at the site.
NB: If you suspect a rare or serious side effect, it is strongly recommended to seek medical advice either from a GP or the medical clinic where the BCG was administered. For specialist immunisation advice or to report an adverse event following immunisation (AEFI), please contact SAEFVIC.
Post vaccination care
Following vaccination the site must be kept clean and dry. Normal activities like bathing and swimming can continue ensuring that the area is patted dry afterwards.
If the ulcer is oozing it may be covered with a dry gauze and can be cleaned with an alcohol swab. Ointments, creams or antiseptics should not be applied directly to the site.
For more information refer to What to expect following the BCG vaccination – RCH parent handout.
Resources
BCG Clinics in Victoria
- The Royal Children’s Hospital: BCG Clinic
- Monash Health Immunisation Service
- Offspring Child Health Specialists
- Family Immunisation & Travel Specialists (FITS)
- A/Prof Mike Starr
- Craigieburn Specialist Consulting Suites
- Medical One- QV
- Kids Travel Doc
- The Children’s Private Medical Group
- Wyndham Private Medical Centre
- Travel Clinics Australia- Caulfied
- Pinnacle Medical Centre – Williams Landing
- The Travel Doctor – TMVC at Sonic HealthPlus CBD clinic
- CBD Doctors Melbourne
Other
- MVEC: Intradermal vaccination
- RCH Kids Health Information: Tuberculosis disease (TB)
- Australian Immunisation Handbook: Tuberculosis
- RCH Kids Health Information BCG vaccine for TB
- What to expect following the BCG vaccination – RCH parent handout
- What to expect after your child receives the BCG vaccination – Monash Hospital parent handout
- Dhanawade, S. Kumbhar, S. Gore, A. and Patil, V. Scar formation and tuberculin conversion following BCG vaccination in infants: A prospective cohort study, Journal of Family Medicine and Primary Care 2015 Jul-Sep 4(3) 384-387
Authors: Nigel Crawford (Director SAEFVIC, Murdoch Children’s Research Institute) and Rachael McGuire (SAEFVIC Research Nurse, Murdoch Children’s Research Institute)
Reviewed by: Rachael McGuire (MVEC Education Nurse Coordinator)
Date: March 13, 2024
Materials in this section are updated as new information and vaccines become available. The Melbourne Vaccine Education Centre (MVEC) staff regularly reviews materials for accuracy.
You should not consider the information in this site to be specific, professional medical advice for your personal health or for your family’s personal health. For medical concerns, including decisions about vaccinations, medications and other treatments, you should always consult a healthcare professional.